Radioactive Half-Life
Concept and Calculation
Radioactive decay is the process by which unstable nuclei emit particles or energy to reach a more stable configuration. The half-life of a radioactive isotope is the time it takes for exactly half the atoms in a sample to decay. This value is a fixed property of each isotope and doesn't change based on how much material you have, what temperature it's at, or any other external condition.
The relationship between half-life and the decay constant (which describes the probability of decay per unit time) is:
To find how many radioactive nuclei remain after some elapsed time :
where is the initial number of radioactive nuclei.
Half-lives span an enormous range depending on the isotope:
- Carbon-14: ~5,730 years (useful for dating organic remains)
- Uranium-238: ~4.5 billion years (useful for dating the age of rocks and the Earth itself)
- Technetium-99m: ~6 hours (useful in medical imaging because it decays quickly and doesn't linger in the body)
Applications and Significance
The predictable nature of radioactive decay makes it useful across many fields:
- Geological and archaeological dating: Decay acts as a natural clock. By measuring how much of a parent isotope has converted to its daughter product, scientists can determine the age of a sample.
- Medical imaging: Short half-life isotopes like technetium-99m (~6 hours) deliver diagnostic information without prolonged radiation exposure.
- Cancer treatment: Isotopes like iodine-131 (~8 day half-life) are chosen so they deliver therapeutic radiation over a controlled window of time.
- Environmental tracing: Tritium (~12.3 year half-life) is used in hydrology to track water movement through systems.
- Nuclear power: Reactors harness energy from fission of long-lived isotopes like uranium-235 (~700 million year half-life).
Exponential Radioactive Decay

Mathematical Representation
Radioactive decay is exponential because the rate of decay at any moment is proportional to how many radioactive nuclei are present right then. More undecayed atoms means more decays per second; as the sample shrinks, the decay rate drops.
The exponential decay law is:
where is the number of radioactive nuclei at time , is the initial count, and is the decay constant. You can convert between and half-life with:
The activity of a sample (the number of decays per unit time, measured in becquerels or curies) follows the same exponential pattern:
If you graph vs. time, you get a curve that drops steeply at first, then gradually flattens as it approaches zero without ever quite reaching it. This shape reflects the probabilistic nature of decay: each individual nucleus has a fixed probability of decaying in any given time interval, but you can never guarantee that every last one has decayed.
Practical Implications
- Waste management: Exponential decay means radioactive waste never technically reaches zero activity. In practice, facilities plan storage times based on how many half-lives are needed to reduce activity to safe levels (often 10+ half-lives).
- Biological systems: In the body, a radioactive substance is removed by both physical decay and biological processes (excretion, metabolism). The effective half-life combines both: . This is why medical isotopes clear the body faster than their physical half-life alone would suggest.
- Safety protocols: Shielding requirements and handling procedures are designed around the expected activity at each point in time.
- Detector design: Instruments must account for the decreasing signal strength as a source decays, especially during long measurements.
Radioactive Dating Techniques
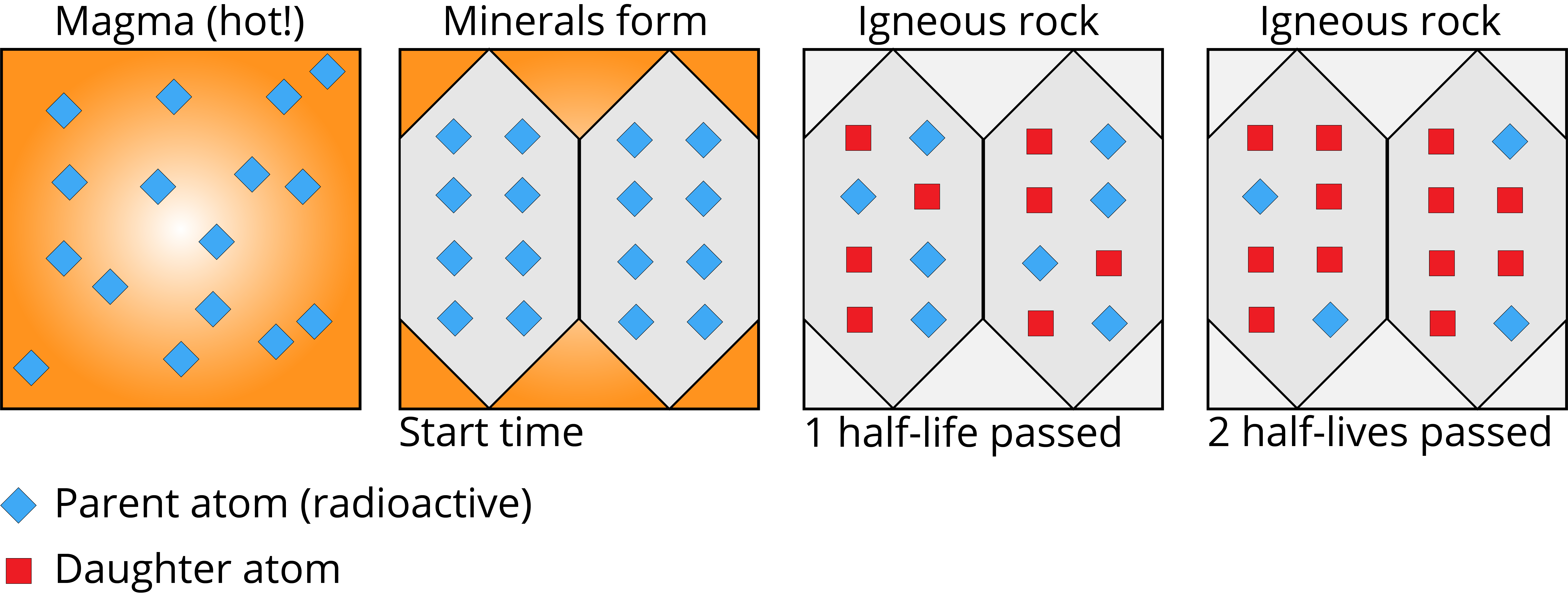
Common Methods and Applications
Different isotope systems are suited to different age ranges and material types. The general idea is always the same: measure the ratio of parent isotope to daughter isotope, then use the known decay rate to calculate how much time has passed.
The age equation is:
where is the number of daughter atoms, is the number of remaining parent atoms, and is the decay constant of the parent.
Here are the most commonly used systems:
| Method | Parent → Daughter | Half-Life | Typical Use |
|---|---|---|---|
| Carbon-14 | ~5,730 years | Organic materials up to ~50,000 years old | |
| Potassium-Argon | ~1.25 billion years | Volcanic rocks, older geological samples | |
| Uranium-Lead | ~4.5 billion years | Very old rocks and minerals | |
| Rubidium-Strontium | ~48.8 billion years | Ancient igneous and metamorphic rocks | |
| Thorium-Lead | ~14 billion years | Thorium-rich minerals |
Notice the pattern: short half-life isotopes (like carbon-14) are good for young samples, while long half-life isotopes are needed for ancient ones. If you tried to use carbon-14 on a billion-year-old rock, essentially all the would be gone and there'd be nothing left to measure.
Principles and Procedures
The general workflow for radiometric dating:
- Select the right isotope system based on the expected age range and the type of material (organic vs. mineral, for example).
- Collect and prepare the sample carefully to avoid contamination from external sources of parent or daughter isotopes.
- Measure the parent-to-daughter ratio using mass spectrometry or another high-sensitivity technique.
- Apply calibration corrections where needed. For carbon-14 dating, calibration curves account for the fact that atmospheric production has varied over time due to changes in cosmic ray flux and Earth's magnetic field.
- Cross-validate using multiple dating methods when possible to increase confidence.
- Consider geological context, such as stratigraphic position (which layers are above or below), to check whether the radiometric age makes sense.
Radioactive Dating Limitations
Assumptions and Potential Errors
Every radiometric dating method rests on a few key assumptions. When these assumptions break down, errors creep in.
- Known initial conditions: You need to know (or reliably estimate) how much daughter isotope was present when the sample formed. If the starting ratio is wrong, the calculated age will be wrong.
- Closed system: The method assumes no parent or daughter isotopes have been added or removed since the sample formed. Weathering, heating, or fluid flow through rock can violate this assumption.
- Constant decay rate: Decay constants are treated as truly constant over time. Extensive experimental evidence supports this, but it's still a foundational assumption.
- Cosmic ray variations: For carbon-14 specifically, changes in cosmic ray intensity over time alter the production rate of in the atmosphere. Without calibration, this introduces systematic errors.
- Measurement precision: The statistical nature of radioactive decay and the sensitivity limits of instruments both constrain precision. Very old samples (little parent left) and very young samples (little daughter accumulated) are the hardest to date accurately.
- Age range limits: Each method has a practical window. Carbon-14 dating can't reliably go beyond ~50,000 years because too little remains to measure.
Mitigation Strategies and Considerations
- Multiple methods: Using two or more independent dating techniques on the same sample helps identify discrepancies and builds confidence in the result.
- Isochron dating: This technique plots data from multiple minerals in the same rock to determine both the age and the initial daughter isotope ratio simultaneously, reducing dependence on assumptions about initial conditions.
- Improved calibration: Dendrochronology (tree-ring counting), ice cores, and coral records help refine carbon-14 calibration curves going back tens of thousands of years.
- Better sample preparation: Advances in chemical cleaning and separation techniques reduce contamination. Accelerator mass spectrometry (AMS) allows dating of much smaller samples, making it easier to select uncontaminated material.
- Geological cross-checks: Stratigraphic relationships, paleomagnetism, and fossil assemblages provide independent age constraints that can confirm or challenge radiometric results.
- Reporting uncertainties: Responsible dating always includes error bars (e.g., years). These reflect both measurement uncertainty and known sources of systematic error.