Electric Potential and Electric Potential Energy
Electric potential vs. potential energy
These two terms sound similar but describe different things. Understanding the distinction is critical for solving problems in this unit.
Electric potential (also called voltage) is a scalar quantity measured in volts (V). It describes the electric potential energy per unit charge at a point in an electric field. Think of it as a property of a location in space, regardless of what charge you place there.
Electric potential energy is a scalar quantity measured in joules (J). It's the actual energy stored in a system due to the arrangement of charges. Unlike potential, it depends on both the voltage and the amount of charge present.
A helpful comparison: electric potential is like the height of a hill, while electric potential energy is like the gravitational potential energy of a specific boulder sitting on that hill. The hill's height doesn't change, but the energy depends on how massive the boulder is.
Potential difference and energy relationship
The connection between potential difference and energy is given by:
- : change in electric potential energy (J)
- : charge (C)
- : potential difference, or voltage difference between two points (V)
This equation tells you that moving a larger charge through a given potential difference results in a larger change in energy. Similarly, a bigger potential difference means more energy transferred for the same charge.
Practical scenarios:
- Batteries create a fixed potential difference between their terminals. A 9 V battery does more work on a given charge than a 1.5 V battery because is larger.
- Capacitors store electric potential energy in the electric field between two conducting plates. The potential difference across the plates determines how much energy is stored.
Equipotential surfaces are regions where the electric potential has the same value everywhere. Moving a charge along an equipotential surface requires zero work, because and therefore .

The electron volt
The joule is a convenient unit for everyday energy, but it's far too large for atomic-scale processes. The electron volt (eV) fills that gap.
One electron volt is the energy gained (or lost) when a single electron moves through a potential difference of 1 volt:
You can verify this using : the charge of an electron is C, and V, so the energy is J.
Where electron volts show up:
- Atomic binding energies: The energy holding an electron in a hydrogen atom's ground state is 13.6 eV. Writing that as J would be awkward.
- Photon energies: Using , visible light photons have energies of roughly 1.8 to 3.1 eV, while X-ray photons reach into the keV range.
- Particle accelerators: Energies are expressed in MeV ( eV) or GeV ( eV). The Large Hadron Collider operates at energies of several TeV ( eV) per beam.
Calculations with potential difference
The core formula is the same one from above:
Here's a step-by-step approach for solving problems:
- List what you know. Identify the charge and the potential difference (or whichever two of the three quantities are given).
- Identify what you're solving for. Usually it's , but you might need to find or instead.
- Substitute and solve. Plug the known values into and solve for the unknown.
- Check units. Coulombs times volts gives joules. If the problem asks for eV, divide your answer in joules by .
Example: A proton ( C) moves through a potential difference of 500 V. What is its change in potential energy?
In electron volts: (since a proton has the same magnitude of charge as an electron, moving it through 500 V gives 500 eV).
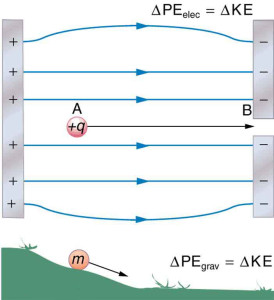
Energy and work in electric fields
Electric fields exert forces on charges, and those forces can do work. When the electric field does positive work on a charge, the charge's electric potential energy decreases and its kinetic energy increases, just like gravity does positive work on a falling ball.
Conservation of energy still applies:
A positive charge released from rest in a uniform electric field will accelerate in the direction of the field, converting potential energy into kinetic energy. This principle is exactly how particle accelerators work: charges gain kinetic energy by moving through large potential differences.