Lipids are a broad class of biomolecules united by one key trait: they don't dissolve well in water. This hydrophobic character underlies everything lipids do, from storing energy in fat tissue to forming the membranes that define every cell. Understanding the structural differences between lipid types helps explain why each one functions the way it does.
Triglycerides, phospholipids, and steroids are the three main lipid categories you need to know. Fatty acids and glycerol serve as the building blocks for many of them, and variations in fatty acid structure (saturated vs. unsaturated) directly affect a lipid's physical properties.
Types and Structures of Lipids
Classify the main types of lipids, including triglycerides, phospholipids, and steroids
Triglycerides consist of three fatty acid chains bonded to a single glycerol backbone through ester bonds (a reaction called dehydration synthesis). They function as the body's primary long-term energy storage molecule. You encounter triglycerides as fats (solid at room temperature, mostly saturated) and oils (liquid at room temperature, mostly unsaturated).
Phospholipids look similar to triglycerides but with one key difference: one of the three fatty acid positions is replaced by a phosphate group, often linked to an additional small molecule. This gives them an amphipathic structure, meaning one end is hydrophilic (the phosphate head) and the other end is hydrophobic (the two fatty acid tails). That dual nature is what allows phospholipids to spontaneously form bilayers in water. Examples include lecithin and sphingomyelin.
Steroids have a completely different architecture: four fused carbon rings rather than long fatty acid chains. Cholesterol is the most well-known steroid. It sits within cell membranes to modulate fluidity, and it also serves as the precursor molecule for steroid hormones (like testosterone and estrogen) and bile acids used in digestion.
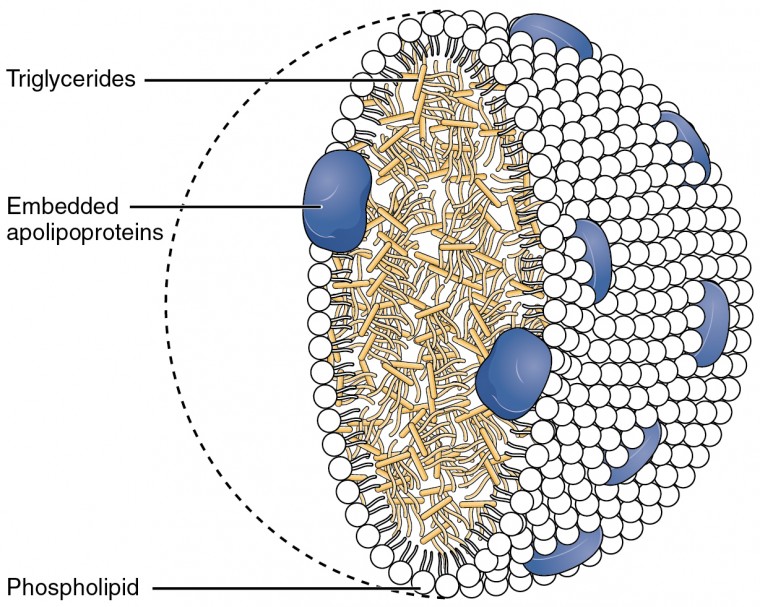
Describe the structural components of lipids, such as fatty acids and glycerol
Fatty acids are long hydrocarbon chains with a carboxyl group () at one end. The hydrocarbon chain is what makes them hydrophobic. Two important variations:
- Saturated fatty acids have only single bonds between carbons. The chains pack tightly together, which is why saturated fats (like palmitic acid and stearic acid) tend to be solid at room temperature.
- Unsaturated fatty acids contain one or more carbon-carbon double bonds, which introduce kinks in the chain. These kinks prevent tight packing, so unsaturated fats (like oleic acid and linoleic acid) are usually liquid at room temperature.
Glycerol is a simple three-carbon molecule with a hydroxyl group () on each carbon. It acts as the backbone for both triglycerides and phospholipids. Each hydroxyl group can form an ester bond with a fatty acid's carboxyl group (releasing water), or in the case of phospholipids, one hydroxyl bonds to a phosphate group instead.

Functions and Properties of Lipids
Explain the functions of lipids in living organisms, including energy storage, cell membrane structure, and signaling
Energy storage. Triglycerides are extremely efficient energy reserves. Gram for gram, fats store about twice as much energy as carbohydrates because their hydrocarbon chains are highly reduced (lots of C-H bonds to oxidize). Adipose tissue stores triglycerides and also provides insulation and cushioning for internal organs.
Cell membrane structure. Phospholipid bilayers form the structural foundation of all cell membranes. The hydrophobic interior of the bilayer acts as a barrier that prevents most polar molecules and ions from freely crossing. Proteins embedded in or attached to the bilayer handle selective transport (ion channels, carrier proteins) and cell communication (receptor proteins).
Signaling. Several lipid types function as signaling molecules:
- Steroid hormones like testosterone and cortisol pass through cell membranes (they're hydrophobic), bind to intracellular receptors, and directly regulate gene expression.
- Membrane phospholipids can be cleaved to generate second messengers such as diacylglycerol (DAG) and inositol trisphosphate (). These activate downstream targets like protein kinase C (PKC) and trigger calcium release from intracellular stores.
Discuss the properties of lipids, such as hydrophobicity and their role in the formation of biological membranes
The defining property of lipids is hydrophobicity, which comes from their long nonpolar hydrocarbon chains. In an aqueous environment, hydrophobic molecules are pushed together by water (this is driven by entropy, not by attraction between the lipids themselves). This tendency causes lipids to aggregate into structures like micelles and bilayers.
Biological membrane formation follows a predictable sequence:
- Phospholipids placed in water spontaneously arrange so that hydrophilic heads face the aqueous environment and hydrophobic tails face inward, away from water.
- This self-assembly produces a bilayer that acts as a selective barrier to polar molecules and ions.
- The fluidity of the resulting membrane depends on fatty acid composition:
- Shorter chains and more unsaturated (kinked) fatty acids increase fluidity by preventing tight packing.
- Longer chains and more saturated (straight) fatty acids decrease fluidity by allowing tighter packing.
- Cholesterol acts as a fluidity buffer: at high temperatures it reduces fluidity, and at low temperatures it prevents the membrane from becoming too rigid.
Cholesterol's buffering role is a common exam topic. It doesn't simply increase or decrease fluidity; it stabilizes the membrane across a range of temperatures.