What is Cell Potential?
In the previous section, we explored the idea of electromotive force from redox reactions! Electromotive force is the “pull” on electrons from a reducing agent to an oxidizing agent. The higher the electromotive force is, the more spontaneous the reaction will be.
We can conceptualize electromotive force by generally thinking about how strongly each chemical species wants to either get rid of electrons or gain electrons. Although narrowing the scope to atoms or ions exchanging electrons this way is not always the most realistic representation, it can help us to visualize how electromotive force may work. Electromotive force, in units of volts, is a way to measure cell potential.
When dealing with a galvanic cell, the reduction and the oxidation occur on separate sides. The anode is the side where oxidation occurs, and the cathode is where reduction occurs. (Remember: An Ox; Red Cat)
Electrons travel through a wire from the anode to the cathode. The force these electrons experience as they pull through the wire is known as the cell potential. It is the same as the electromotive force caused by the redox reaction.
If a cell is under standard conditions, that means that the cell has 1 M concentrations and is at a temperature of 298 K and a pressure of 1 atm. We can calculate standard cell potential using the equation E°cell = Ecathode - Eanode. In other words, the standard cell potential for a cell is the reduction potential for the species in the cathode minus the reduction potential of the species in the anode.
Take a look at this example:
Calculate the cell potential for the following cell given the electrochemical data below:
2AgBr + 2Hg → 2Ag + Hg2Br2
Hg2Br2 + 2e- → 2Hg + 2Br- (E = +0.140 V)
2AgBr + 2e- → 2Ag + 2Br- (E = +0.071 V)
For this reaction, we see that AgBr reduces into Ag and Br-, so the cathode will be AgBr. At the anode, Hg oxidizes.
Plugging into our equation, we find that for E°cell = Ecathode - Eanode, E°cell = 0.071 V - 0.140 V = -0.069 V.
Another strategy for finding cell potential is to negate the oxidation reaction and then add the cell potentials together. For example, looking at the previous question, we could have also acknowledged that the actual half-reaction for Hg was 2Hg + 2Br- → Hg2Br2 + 2e-, which has a potential of -0.140 V. If we do E°cell = +0.071 V + (-0.140 V), the math yields the same result of -0.069 V.

Calculating Cell Potential Using Reduction Potentials
In the last problem, we were given specific electrochemical data, but for some questions, you will need to reference and apply values from a table of standard reduction potentials. Essentially, a table of standard reduction potentials is a very long list of reduction reactions with their corresponding potentials.
To find the correct value requires the right set of half-reactions, including which half-reaction occurs at the anode and which occurs at the cathode. Once we make these determinations, we can apply our cell potential equation: E°cell = Ecathode - Eanode.
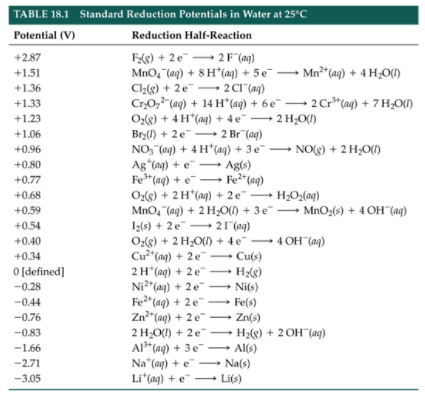
The AP Exam will directly provide the necessary data to solve for cell potential. However, familiarity with a table of standard reduction potentials helps to build conceptual understanding, which is useful in practice and on testing day. Below is a sample table of standard reduction potentials:
Image From Grade12UChemNote that the reduction of H+ into H2 is a defined value of 0 V. This is because all other reduction potentials are modeled by comparing their voltage to the voltage of this reduction, essentially asking the question, “is this reaction more spontaneous or less spontaneous than 2H+ + 2e- → H2?”
The values in this table are all reductions, which is why we negate the potential if we manipulate the reactions to be oxidations. For many substances, the reduction has a negative potential. What does that mean? A negative reduction potential indicates that species like sodium (Na) and lithium (Li) are excellent reducing agents rather than species that are easy to reduce. If we flip the reductions in these cases, we see the negative reduction potentials become positive in the instance of oxidation, meaning oxidation is more favorable.
Standard Cell Potentials and Spontaneity 🔋
The major conclusion we want to draw from finding cell potential is the spontaneity and thermodynamic favorability of the redox reaction taking place. If E°cell is positive, the reaction is spontaneous. Why? The electromotive force is strong enough to pull the electrons off the reducing agent and onto the oxidizing agent. Conversely, if the E°cell is negative, the reaction is nonspontaneous. Therefore, we can predict the sign on ΔG° for a reaction given E°cell.
To reiterate:
If E°cell > 0, the reaction is spontaneous (thermodynamically favored), and ΔG° is negative.
If E°cell < 0, the reaction is nonspontaneous (thermodynamically unfavored), and ΔG° is positive.
Calculating ΔG° Using E°cell
While we can look at the sign on E°cell to predict the sign of ΔG°, we also have a way of directly calculating ΔG° from cell potential. The formula for ΔG° using E°cell is as follows:
ΔG° = -nFE°cell
In this equation, ΔG° is the standard Gibbs Free Energy change, E°cell is the standard cell potential, n is the number of moles of electrons transferred in the reaction, and F is Faraday’s constant, which is 96,485 coulombs/mol e-. A coulomb is a measure of electric charge.
We can take a look at the following example:
Suppose we have a galvanic cell for which E°cell = 1.02 V. In the reaction that occurs, 1 mol of electrons transfer. At 298K, what is ΔG° for this cell? What is the equilibrium constant, K?
First we can plug into ΔG° = -nFE°cell to find ΔG°:
ΔG° = -nFE°cell
ΔG° = -1(96,485)(1.02) = -98414.7 J = -98.414 kJ
Therefore ΔG° = -98414.7 J or -98.414 kJ.
Next, we can use our relationship between ΔG° and K to find the equilibrium constant:
K = e^(-ΔG°/RT)
K = e^(98414.7 /(8.314)(298)) = 1.78 * 10^17
Congratulations! Now you know how to calculate cell potential and how to apply cell potentials to calculate the standard change in Gibbs Free Energy.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
anode | The electrode where oxidation occurs in an electrochemical cell. |
cathode | The electrode where reduction occurs in an electrochemical cell. |
current | The flow of electrical charge through a circuit, measured in amperes. |
electrochemical cell | A device consisting of electrodes and electrolyte solutions where redox reactions occur, either spontaneously (galvanic) or non-spontaneously (electrolytic). |
electrode | A conductor through which electric current enters or leaves an electrochemical cell. |
electrode mass | The mass of an electrode, which can change during electrochemical reactions due to oxidation or reduction at the electrode surface. |
electrolytic cell | An electrochemical cell in which electrical energy is used to drive a non-spontaneous redox reaction. |
electron flow | The movement of electrons through the external circuit of an electrochemical cell from the anode to the cathode. |
galvanic cell | An electrochemical cell in which a spontaneous redox reaction generates electrical current. |
half-cells | Individual compartments of an electrochemical cell, each containing an electrode and electrolyte solution where a half-reaction occurs. |
half-reaction | Separate equations showing either the oxidation process (loss of electrons) or the reduction process (gain of electrons) in a redox reaction. |
ion flow | The movement of ions through the electrolyte and salt bridge to complete the circuit in an electrochemical cell. |
oxidation | The loss of electrons by a substance in a redox reaction. |
reduction | The gain of electrons by a substance in a redox reaction. |
salt bridge | A tube containing an inert electrolyte that connects two half-cells and allows ion flow to maintain electrical neutrality. |
thermodynamically favored | A reaction or process that has a negative Gibbs free energy (ΔG < 0) and is spontaneous under given conditions. |
thermodynamically unfavored | A reaction that does not proceed spontaneously under standard conditions, resulting in a negative cell potential and positive Gibbs free energy change. |
Frequently Asked Questions
What is cell potential and how does it relate to free energy?
Cell potential (E°cell) is the voltage produced by a redox cell under standard conditions. You find it by adding the standard reduction potentials of the two half-reactions after making one an oxidation (flip sign). If E°cell is positive the reaction is thermodynamically favored (galvanic); if negative it’s unfavored and needs an external potential (electrolytic). Free energy and cell potential are directly related by ΔG° = −nFE°. Here n = moles of electrons transferred, F ≈ 96,485 C·mol⁻¹ (Faraday constant), and E° is in volts; ΔG° comes out in joules (divide by 1000 for kJ). So positive E° → negative ΔG° → spontaneous. For nonstandard conditions use the Nernst equation to find E and then ΔG = −nFE. This aligns with CED 9.9.A (standard potentials and spontaneity). For a focused review see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why does a positive cell potential mean the reaction is spontaneous?
A positive standard cell potential (E°cell > 0) means the redox reaction is spontaneous because E° and Gibbs free energy (ΔG°) are directly linked by ΔG° = -nFE°. Here n is the number of electrons transferred and F is Faraday’s constant (~96,485 C·mol⁻¹). If E° is positive, -nFE° is negative, so ΔG° < 0—that’s the thermodynamic criterion for spontaneity. In electrochemical terms a positive E° means the reduction half-reaction chosen as the cell’s cathode has a higher tendency to be reduced than the oxidation half-reaction has to be reduced, so electrons flow spontaneously from anode to cathode (a galvanic cell). On the AP exam you should be able to (a) compute E° from standard reduction potentials, (b) use ΔG° = -nFE° to decide spontaneity, and (c) relate sign of E° to galvanic vs. electrolytic behavior (CED 9.9.A). For a nonstandard cell, use the Nernst equation to get E and then ΔG = -nFE to judge spontaneity. For review, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and the Unit 9 page (https://library.fiveable.me/ap-chemistry/unit-9). For practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about the difference between standard reduction potential and standard cell potential - can someone explain?
Standard reduction potential (E°red) is the voltage for one half-reaction written as a reduction under standard conditions (1 M, 1 atm, 25°C). It’s a property of a single electrode (e.g., Ag+ + e– → Ag has E°red = +0.80 V). Standard cell potential (E°cell) is the net voltage for a full electrochemical cell made by pairing two half-reactions. You get it by identifying which half-reaction is reduced (cathode) and which is oxidized (anode) and using: E°cell = E°(cathode, reduction) – E°(anode, reduction). So you subtract the anode’s standard reduction potential (you don’t need to flip signs for electrons; just choose cathode minus anode). Sign meaning: E°cell > 0 → spontaneous galvanic cell (thermodynamically favored per 9.9.A; ΔG° = –nFE°), E°cell < 0 → nonspontaneous (needs external potential). Remember ΔG° = –nFE° (n = electrons; F = Faraday constant) when connecting potentials to free energy on AP problems (Topic 9.9). For a focused review and worked examples, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb); for broader Unit 9 review see (https://library.fiveable.me/ap-chemistry/unit-9) and for practice problems check (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate the standard cell potential from half-reactions?
Pick the two standard reduction half-reactions and their E°(reduction) values. The half-reaction with the more positive E° is the reduction (cathode); the other is reversed to be the oxidation (anode). Then calculate: E°cell = E°(cathode, reduction) − E°(anode, reduction) (Equivalently, E°cell = E°red(cathode) + E°ox(anode) if you convert the anode to an oxidation potential.) If E°cell > 0 the cell reaction is thermodynamically favored (galvanic); if E°cell < 0 it’s unfavored (electrolytic). Use ΔG° = −nFE° to connect to free energy (n = electrons transferred, F ≈ 96485 C·mol−1). This procedure matches the CED essential knowledge 9.9.A.2–3. For worked examples and AP-style practice, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and the AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when ΔG° = -nFE° and how do I use this equation?
ΔG° = -nFE° connects thermodynamics to electrochemistry. It tells you the standard Gibbs free energy change (ΔG°) for a redox reaction from the standard cell potential (E°). What each symbol means: - ΔG°: standard free energy change (J/mol)—negative = spontaneous. - n: number of electrons transferred in the balanced redox reaction. - F: Faraday constant ≈ 96,485 C·mol⁻¹ (charge per mole of electrons). - E°: standard cell potential (V). How to use it: 1. Write and balance the overall cell reaction, find n (total e⁻ transferred). 2. Calculate E° by combining standard reduction potentials (E°cell = E°red(cathode) − E°red(anode)). 3. Plug into ΔG° = -nFE°. Units: V × C = J, so ΔG° will be in joules per mole of reaction. Convert to kJ if wanted. Quick example: E° = 1.10 V, n = 2 → ΔG° = −2(96,485)(1.10) ≈ −2.12×10^5 J ≈ −212 kJ (spontaneous). On the AP exam you may be asked to decide spontaneity from E° or compute ΔG°; positive E° ⇒ ΔG° negative ⇒ thermodynamically favored (CED 9.9.A). For extra review see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why is there a negative sign in the equation ΔG° = -nFE°?
The negative sign comes from how Gibbs free energy and electrical work are defined. For an electrochemical cell, the electrical work done by the reaction when n moles of electrons move is w_elec = -nFE (work done by the system is negative in thermodynamics). The maximum non-PV work the reaction can do equals –ΔG, so ΔG° = -w_max = - ( -nFE° ) → ΔG° = -nFE°. Practically that means: a positive standard cell potential (E° > 0) gives ΔG° < 0, so the redox reaction is thermodynamically favored (spontaneous). Here F is Faraday’s constant (≈ 96,485 C·mol⁻¹) and n is electrons transferred. This sign convention links CED Essential Knowledge 9.9.A.3: E° positive → spontaneous (ΔG° negative). For a quick review, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between thermodynamically favored and thermodynamically unfavored electrochemical cells?
Thermodynamically favored cells: E°cell > 0 so ΔG° = −nFE° is negative → reaction is spontaneous (you can get electrical work; this is a galvanic/voltaic cell). Thermodynamically unfavored cells: E°cell < 0 so ΔG° is positive → reaction isn’t spontaneous and requires an external applied potential to run (an electrolytic cell). How you tell: write the two half-reactions, pick the one with the higher (more positive) standard reduction potential as the cathode (reduction) and the other as the anode (oxidation). E°cell = E°(cathode) − E°(anode) (or sum the appropriate reduction/oxidation values). Then use ΔG° = −nFE° (n = electrons transferred, F = Faraday constant) to check sign/magnitude. For AP prep, be ready to identify half-reactions, calculate E°cell from standard potentials, and link sign of E° to spontaneity on free-response or multiple-choice (see Topic 9.9 study guide for a focused review: https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb). For more practice problems, visit https://library.fiveable.me/practice/ap-chemistry.
How do I know which half-reaction is oxidation and which is reduction when calculating cell potential?
Look at the standard reduction potentials for both half-reactions. The half-reaction with the more positive E°(reduction) will be reduced (cathode); the one with the less positive (more negative) E°(reduction) will be oxidized (anode). Then calculate E°cell as E°cell = E°(cathode, reduction) − E°(anode, reduction). If E°cell > 0 the cell reaction is thermodynamically favored (galvanic); if E°cell < 0 the reaction is nonspontaneous as written (electrolytic). Quick checklist for AP problems: write both reactions as reductions, compare E° values, pick the larger E° as the reduction, flip the other to oxidation when writing the full cell reaction, and use E°cell = E°red(cathode) − E°red(anode). Practice this with the Ag/Cr example in the CED (question 12). For a short refresher, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and do more problems at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain why negative cell potential means you need external energy for the reaction to happen?
A negative standard cell potential (E°cell < 0) means the reaction is thermodynamically unfavored because ΔG° and E° are directly related by ΔG° = -nFE° (n = electrons transferred, F ≈ 96,485 C·mol⁻¹). If E°cell is negative, then -nFE° is positive → ΔG° > 0, so the process is nonspontaneous and will not do useful electrical work on its own. Practically: a galvanic (voltaic) cell has E° > 0 and generates current spontaneously; an electrolytic cell has E° < 0 and requires an external voltage larger than |E°cell| to drive the reaction. For AP wording, use CED 9.9.A: positive E° → thermodynamically favored; negative E° → requires an applied potential (external energy) to proceed. For more practice and a clear walkthrough, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and try related problems at Fiveable practice (https://library.fiveable.me/practice/ap-chemistry).
What does the n represent in the equation ΔG° = -nFE° and how do I find it?
n is the number of moles of electrons transferred in the overall balanced redox reaction. To find n: write the two half-reactions (one oxidation, one reduction), balance them for electrons, then add them so the electrons cancel—the total electrons moved (per formula unit of reaction) is n. Example: Cu2+ + 2 e- → Cu and Zn → Zn2+ + 2 e- gives n = 2 for Zn + Cu2+ → Zn2+ + Cu. In the equation ΔG° = −n F E°, F is the Faraday constant (≈ 96,485 C·mol−1 e−) and E° is in volts (1 V = 1 J·C−1). Using n ensures units match so ΔG° comes out in joules (or convert to kJ). For AP exam practice, be ready to identify half-reactions from cell notation and use the standard reduction potentials to get E° (CED 9.9.A). For a quick review, see the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how to use standard reduction potentials to predict if a reaction will be spontaneous.
Start by writing the two half-reactions and their standard reduction potentials (E°red). The cathode is the half-reaction with the larger E°red (it’s reduced); the anode is the one with the smaller E°red (it’s oxidized). You don’t change the E° value when you flip a half-reaction to oxidation—you only reverse the reaction. Calculate the standard cell potential: E°cell = E°red(cathode) − E°red(anode). If E°cell > 0 the cell is spontaneous (galvanic); if E°cell < 0 it’s nonspontaneous (electrolytic). Use ΔG° = −nFE° to connect to free energy: positive E° gives ΔG° < 0 (thermodynamically favored), negative E° gives ΔG° > 0. For nonstandard conditions use the Nernst equation to adjust E. This is exactly what AP CED 9.9.A expects—identify half-reactions, compute E°cell, and link sign of E° to spontaneity (and ΔG°) (see topic study guide: https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb; unit overview: https://library.fiveable.me/ap-chemistry/unit-9). For extra practice, try problems at Fiveable (https://library.fiveable.me/practice/ap-chemistry).
Why do we study electrochemistry and how does it connect to thermodynamics?
We study electrochemistry because redox reactions in cells let you convert chemical energy to electrical work—and that’s just thermodynamics in action. The standard cell potential E° tells you whether a redox process is thermodynamically favored: E° > 0 for a spontaneous (galvanic) cell, E° < 0 for a nonspontaneous (electrolytic) process. Use standard reduction potentials for each half-reaction to get E°cell, then relate it to free energy with ΔG° = −nFE° (n = electrons transferred, F ≈ 96485 C·mol-1). A positive E° gives a negative ΔG° (spontaneous) and vice versa. The Nernst equation extends this to nonstandard conditions and links cell voltage to reaction Q and equilibrium (useful for predicting direction and equilibrium position). For AP exam work, Learning Objective 9.9.A expects you to identify half-reactions, calculate E°, and state spontaneity (practice these skills in the Topic 9.9 study guide (https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb) and more problems at (https://library.fiveable.me/practice/ap-chemistry)).
How do you determine if an electrochemical cell will produce electricity or need electricity input?
Check the sign of the standard cell potential, E°. Build the cell by picking the reduction half-reaction (higher E°) and the oxidation half-reaction (reverse the lower E° reduction). E°cell = E°(cathode) − E°(anode). If E°cell > 0 the reaction is thermodynamically favored (galvanic cell) and will produce electricity; if E°cell < 0 the reaction is thermodynamically unfavored and needs an external potential (electrolytic cell). You can also use ΔG° = −nFE° (n = electrons transferred, F = 96,485 C·mol−1): positive E° gives ΔG° < 0 (spontaneous). Under nonstandard conditions, use the Nernst equation to find E (and check its sign) to decide spontaneity. For AP-style problems you’ll be asked to identify half-cells, calculate E° from tables of standard reduction potentials, and relate E° to ΔG° (see Topic 9.9 study guide for examples: https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb). For practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between Gibbs free energy and whether a battery will work?
If ΔG° = -nFE° (n = electrons transferred, F ≈ 96,485 C·mol⁻¹), then E° and ΔG° tell you if a battery reaction is thermodynamically favored. A positive standard cell potential (E° > 0) gives ΔG° < 0, so the redox reaction is spontaneous and a galvanic cell will produce useful electrical work. If E° < 0, ΔG° > 0 and the reaction is not spontaneous—the cell won’t run on its own and you need an external potential (electrolytic). Under nonstandard conditions use the Nernst equation to get E (and then ΔG = -nFE). For AP exam framing, this is exactly learning objective 9.9.A and the key equation ΔG° = -nFE° (see Topic 9.9 study guide for more: https://library.fiveable.me/ap-chemistry/unit-9/cell-potential-free-energy/study-guide/GLRagoPDoMJ35XxbRbdb). For extra practice, try problems at https://library.fiveable.me/practice/ap-chemistry.