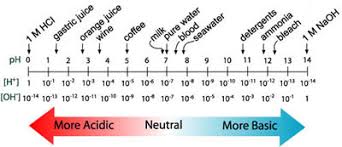
pH and pOH Explained
pH and pOH are extremely important topics in Unit 8 and AP Chemistry as a whole. So then, what is pH? pH is simply a measure of the concentration of protons in a solution.

pH
Essentially, pH is measured by the concentration of H+ ions (or H3O+ ions - they're the same thing in water!), meaning it measures how acidic a solution is. A higher pH means a lower concentration of H+, implying a more basic solution and vice versa (I know, that is really annoying). So, we get why there is an H in pH - H means hydrogen, and pH measures the concentration of hydrogen/hydronium ions. What about the p? In chemistry, 'p' is a symbol that means negative log. p(anything) = -log(anything). Thus, pH = -log([H+]) or pH = -log([H3O+]).
For example, if we had a solution that had 0.01M H+, that would mean pH = -log(1 * 10^-2) = -(-2) = 2 (note that log() implies log base 10).
pOH
If pH = -log([H+]), then one can logically find that pOH = -log([OH-]) and is, in essence, the opposite of pH. Where pH is the concentration of H+, pOH is the concentration of OH-. pOH measures how basic a solution is. A low pOH means a highly basic solution and vice versa.
The Autoionization of Water
To elaborate, let us suggest we had the reaction OH- + H+ --> H2O, which can be described as the protonation of OH- or the autoionization of water. The aforementioned reaction serves as the net ionic for a strong acid-strong base interaction. This reaction can further be described using equilibrium by flipping the reaction to say that H2O ⇌ OH- + H+. The K value for this reaction is a constant called Kw. Kw = 1 * 10^-14.
Think about this reaction a little bit. From Kw that we were given before, we can write that Kw = [OH-][H+]. Therefore, 10^-14 = [OH-][H+]. Now, we can take the negative log of both sides:
14 = -log([OH-][H+]) = -log([OH-]) + -log([H+])
From here, it is clear then that pH + pOH = 14.
pH/pOH of Strong Acid and Strong Base Solutions
Finding the pH of Strong Acid/Base Solutions
For these examples, we will just be looking at pH since it is the most common measure of acidity, but remember, pOH can be easily calculated from this. When dealing with strong acids (and by extension strong bases), what is important is that these reactions go fully forwards. That is to say, they have a K value so high that to call it an equilibrium is negligible. Therefore, all of the acid/base will dissociate.
Take a look at the following example:
Suppose we have a 1M solution of HCl. The dissolution of HCl can be written as: HCl --> H+ + Cl- (or more accurately, HCl + H2O --> H3O+ + Cl-). Because this reaction goes completely forward, we know that 1M of HCl will dissolve into 1M of H+ (or H3O+) and 1M of Cl-. Notice that Cl- is the conjugate base of HCl - when strong acids ionize, they always produce their conjugate base! To find pH, we take the -log([H+]).
pH = -log(1) = -0 = 0. Therefore a 1M solution of HCl has a pH of 0.
The same idea applies to strong bases. Suppose we had 1M NaOH. NaOH --> Na+ + OH-, so pOH = -log([OH-]) = -log(1) = 0.
To find pH, we then plug our value into pH + pOH = 14, which tells us that the pH of a 1M solution of NaOH is 14.
A Special Note About Group II Hydroxides
Here's something important: Group II hydroxides like Ca(OH)₂, Sr(OH)₂, and Ba(OH)₂ are a bit different! When they dissolve, they produce TWO hydroxide ions per formula unit:
Ca(OH)₂ --> Ca²⁺ + 2OH⁻
So if you have a 0.01 M solution of Ca(OH)₂, the [OH⁻] is actually 0.02 M (double the concentration!). This means: pOH = -log(0.02) = 1.70 pH = 14 - 1.70 = 12.30
Always remember to double the OH⁻ concentration for Group II hydroxides!
The List of Strong Acids
There are seven strong acids you need to memorize for AP Chemistry. Luckily, they're not that bad, and they become second nature after a while through practice:
These acids completely ionize in aqueous solutions to produce hydronium ions (H3O+) and their conjugate bases. For example:
- HCl produces Cl⁻ (conjugate base)
- HNO₃ produces NO₃⁻ (conjugate base)
- H₂SO₄ produces HSO₄⁻ (conjugate base) in its first ionization
As such, the concentration of H3O+ in a strong acid solution is equal to the initial concentration of the strong acid, and thus the pH of the strong acid solution is easily calculated.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
complete ionization | The process where all molecules of a strong acid or base separate into ions in aqueous solution. |
conjugate base | The species formed when an acid donates a proton; the base form in an acid-base conjugate pair. |
hydronium ion | The aqueous ion H3O+(aq) formed when a hydrogen ion bonds with a water molecule; represents the form of hydrogen ion in aqueous solution. |
hydroxide ion | The negatively charged ion OH− produced when water autoionizes or when a base dissolves in water. |
ionize | The process by which a compound separates into ions when dissolved in solution. |
pH | A logarithmic scale used to express the concentration of hydronium ions in a solution, calculated as −log[H3O+]. |
pOH | A logarithmic scale used to express the concentration of hydroxide ions in a solution, calculated as −log[OH−]. |
strong acid | An acid that completely dissociates in water and has a very weak conjugate base; examples include HCl, HBr, HI, HClO₄, H₂SO₄, and HNO₃. |
strong base | A base that completely dissociates in water and has a very weak conjugate acid; group I and II hydroxides are common examples. |
Frequently Asked Questions
What is the difference between pH and pOH?
pH and pOH are two ways to report acidity/basicity based on ion concentrations. pH = −log[H3O+] and pOH = −log[OH−]. At 25°C they’re linked by pH + pOH = 14 (because Kw = [H3O+][OH−] = 1.0×10−14). For strong acids/bases (Topic 8.2), use the initial concentration since they completely dissociate: a 0.010 M HCl gives [H3O+] = 0.010 M → pH = 2.0. For bases, group I hydroxides give [OH−] = initial; group II (like Ca(OH)2) give [OH−] = 2×initial. Example: 0.020 M Ca(OH)2 → [OH−]=0.040 M → pOH = 1.40 → pH = 14 − 1.40 = 12.60. The AP CED expects you to calculate pH/pOH from concentrations of all species in strong acid/base solutions (8.2.A). For a quick review, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate pH from molarity of a strong acid?
If the acid is a strong acid it fully ionizes in water, so the hydronium concentration [H3O+] equals the acid’s initial molarity. Then pH = −log10[H3O+]. Steps: 1. Use [H3O+] = initial [acid] for monoprotic strong acids (HCl, HBr, HI, HNO3, HClO4). 2. For strong bases, use [OH−] = initial [base] (Group I) or = 2×[base] (Group II). 3. Calculate pH = −log10[H3O+]. (If you start from [OH−], find pOH = −log10[OH−], then pH = 14.00 − pOH at 25 °C.) Quick example: 0.0200 M HCl → [H3O+] = 0.0200 M → pH = −log(0.0200) = 1.70. Note: H2SO4’s first proton is essentially complete; the second proton may not fully dissociate depending on concentration, so treat carefully. This skill lines up with CED 8.2.A (calculate pH/pOH from concentrations). For more help, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT), the Unit 8 overview (https://library.fiveable.me/ap-chemistry/unit-8), and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do strong acids completely ionize in water?
Strong acids “completely ionize” in water because proton transfer to water is extremely favorable: the acid (HA) gives H+ to H2O to form H3O+ and the conjugate base A−, and that reaction lies almost entirely to the right for strong acids. Physically this happens because (1) water stabilizes the proton as hydronium (solvation/hydration lowers the energy of H3O+), (2) the conjugate bases of strong acids (Cl−, NO3−, ClO4−, etc.) are very stable/weakly basic, and (3) the acid’s equilibrium constant (Ka) is so large that the equilibrium concentration of undissociated HA is negligible. In AP terms (CED 8.2.A.1), [H3O+] ≈ initial [acid], so pH is calculated directly from that concentration (and for strong bases, [OH−] equals the dissolved amount). For practice calculating pH/pOH of strong acids and bases, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about why Group II hydroxides give double the OH- concentration - can someone explain?
Short answer: Group II hydroxides (like Ca(OH)2, Mg(OH)2) have two OH− groups per formula unit. Because they’re strong bases that completely dissociate in water (CED 8.2.A.2), one mole of M(OH)2 yields one mole M2+ and two moles OH−: M(OH)2 → M2+ + 2 OH−. So [OH−] = 2 × [initial M(OH)2]. Quick example: 0.100 M Ca(OH)2 → [OH−] = 0.200 M. Then pOH = −log(0.200) ≈ 0.70 and pH = 14.00 − 0.70 ≈ 13.30. Why that matters for AP: when you calculate pOH/pH for strong bases on the exam, use stoichiometry from the dissociation (not just the initial molarity)—Group I gives [OH−] = initial M, Group II gives double. For more practice and explanations see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and the Unit 8 overview (https://library.fiveable.me/ap-chemistry/unit-8). Need practice problems? Check (https://library.fiveable.me/practice/ap-chemistry).
What are all the strong acids I need to memorize for the AP exam?
Memorize these strong acids for the AP exam: HCl, HBr, HI, HClO4 (perchloric), H2SO4 (sulfuric—note: only the first proton is fully strong), and HNO3 (nitric). The CED lists these as examples you should know for Topic 8.2 because they fully ionize and [H3O+] ≈ initial acid molarity (use that to calculate pH) (see the Topic 8.2 study guide: https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT). For strong bases, memorize group 1 hydroxides (NaOH, KOH, LiOH, etc.) and the soluble group 2 hydroxides that are considered strong (Ca(OH)2, Sr(OH)2, Ba(OH)2). For group I hydroxides [OH–] = initial concentration; for soluble group II hydroxides [OH–] = 2 × initial [base]. These facts are exactly what 8.2.A expects you to use on the exam. For more practice, try problems at Fiveable’s practice page (https://library.fiveable.me/practice/ap-chemistry) and review the Unit 8 overview (https://library.fiveable.me/ap-chemistry/unit-8).
How do you go from pOH to pH using that 14 equation?
That 14 equation is just the relationship pH + pOH = 14 (at 25°C). So to go from pOH to pH: 1. Calculate pOH (for a strong base, pOH = −log[OH−]; for a strong acid you’d get pH directly from [H3O+]). 2. Use pH = 14.00 − pOH. 3. Example: if [OH−] = 1.0×10−3 M, pOH = 3 → pH = 14 − 3 = 11. Note: the “14” comes from Kw = [H3O+][OH−] = 1.0×10−14 at 25°C, so pH + pOH = −log Kw = 14. At other temperatures Kw changes, so the sum won’t be exactly 14. On the AP exam you’ll often use this for strong-acid/base problems (calculate [H3O+] or [OH−], convert to pH or pOH, then use pH + pOH = 14)—see the Topic 8.2 study guide for examples (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT). For more practice questions, check Fiveable’s AP Chemistry practice set (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between strong acids and weak acids in terms of ionization?
Strong acids essentially 100% ionize in water: every molecule donates a proton to form H3O+ and the conjugate base, so [H3O+] equals the initial acid molarity (e.g., 0.010 M HCl → [H3O+] = 0.010 M, pH = 2.00). Weak acids only partially ionize and establish an equilibrium (HA ⇌ H3O+ + A−); you must use an ICE table and Ka to find [H3O+] and percent ionization (e.g., acetic acid). Likewise, strong bases (group I hydroxides and group II hydroxides) completely dissociate: [OH−] = initial [M+] for group I, and = 2 × initial [M2+] for many group II hydroxides (use that to get pOH/pH). These distinctions are exactly what the CED tests in Topic 8.2—you’ll be asked to calculate pH/pOH for strong acids/bases directly, but use equilibrium methods for weak acids (see 8.2.A in the CED). For a quick study refresher, check the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does HCl completely dissociate but other acids don't?
HCl completely dissociates in water because its H–Cl bond is highly polar and relatively weak, and the chloride ion is a very stable, well-solvated conjugate base. That combination makes the forward reaction HCl + H2O → H3O+ + Cl− overwhelmingly favorable (Ka ≫ 1), so essentially every HCl molecule gives H3O+. Other acids don’t fully dissociate because either their H–X bond is stronger or less polar, or their conjugate base is less stable (less able to accept and stabilize the negative charge). That’s the AP idea behind “strong” vs “weak” acids: strong acids (HCl, HBr, HI, HClO4, H2SO4, HNO3) essentially 100% ionize so [H3O+] ≈ initial acid concentration (CED 8.2.A.1). For more practice and quick review of these concepts, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and Unit 8 resources (https://library.fiveable.me/ap-chemistry/unit-8). Want problems to try? Use the AP practice set (https://library.fiveable.me/practice/ap-chemistry).
How do I calculate the pH of 0.01 M NaOH step by step?
Step 1—recognize NaOH is a strong base and fully dissociates: NaOH(aq) → Na+(aq) + OH−(aq). So [OH−] = initial [NaOH] = 0.01 M. Step 2—calculate pOH: pOH = −log[OH−] = −log(0.01) = 2.00. Step 3—convert to pH using Kw at 25°C (pH + pOH = 14): pH = 14.00 − pOH = 14.00 − 2.00 = 12.00. Answer: pH = 12.00 (so the solution is basic). This uses CED Essential Knowledge 8.2.A.2—group I hydroxides fully dissociate. For more worked examples and AP-style practice on Topic 8.2, see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens when you dissolve Ca(OH)2 in water and why does it matter for pH calculations?
Ca(OH)2 is a strong base (a Group II hydroxide) so each formula unit that dissolves gives one Ca2+ and two OH−. That matters for pH because [OH−] = 2 × [Ca(OH)2]dissolved (CED 8.2.A.2). But Ca(OH)2 is only sparingly soluble in water—it will only dissolve until the Ksp equilibrium is reached. Two common AP-style cases: - If you start with a known molar solution (e.g., you prepare 0.10 M Ca(OH)2), assume complete dissociation: [OH−] = 0.20 M → pOH = −log(0.20) ≈ 0.70 → pH ≈ 13.30. - If you dissolve solid in pure water and no concentration is given, use Ksp: Ca(OH)2 ⇌ Ca2+ + 2 OH−, Ksp ≈ 5.5×10−6 = [Ca2+][OH−]^2 = 4s^3, so s ≈ (Ksp/4)1/3 ≈ 0.011 M, [OH−] ≈ 0.022 M → pOH ≈ 1.65 → pH ≈ 12.35. On the AP exam you must state whether to use complete dissociation (given concentration) or a Ksp/saturation calculation (solid in water or past solubility limit). For more examples and practice, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why [H3O+] equals the initial concentration of strong acid - isn't some left unreacted?
Short answer: for a strong acid like HCl the acid molecules essentially all give up their protons to water, so HCl + H2O → H3O+ + Cl− goes to completion. That means the molarity of H3O+ produced equals the initial molarity of the acid (ignoring tiny contributions from water autoionization). Why it feels weird: you’re thinking some acid might “stay” as HA, but by definition a strong acid fully ionizes in water (CED 8.2.A.1). Two quick caveats students see on the AP exam: (1) at very low acid concentrations (≈10−7 M or lower) water’s autoionization matters and you can’t ignore [H3O+] from H2O; (2) polyprotic acids (like H2SO4) may have one dissociation that’s complete and another that isn’t, so watch stoichiometry (CED keywords: complete dissociation, hydronium ion). For more examples and practice on Topic 8.2 see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and tons of practice problems (https://library.fiveable.me/practice/ap-chemistry).
What are the strong bases I need to know and how do they dissociate?
You need to know that “strong bases” on the AP are the soluble Group 1 and the soluble Group 2 hydroxides—they completely dissociate in water, so you can read OH− concentration directly from the initial molarity (CED 8.2.A.2). Examples and dissociation: - Group 1 (one OH per formula unit; [OH−] = initial M): - LiOH → Li+ + OH− - NaOH → Na+ + OH− - KOH → K+ + OH− - RbOH → Rb+ + OH− - CsOH → Cs+ + OH− - Group 2 (two OH− per formula unit; [OH−] = 2 × initial M for soluble ones): - Ca(OH)2 → Ca2+ + 2 OH− - Sr(OH)2 → Sr2+ + 2 OH− - Ba(OH)2 → Ba2+ + 2 OH− (Important: Mg(OH)2 is only sparingly soluble, so it’s not treated as a “completely dissociating” strong base in many cases.) Use [OH−] to find pOH (pOH = −log[OH−]) and then pH (pH + pOH = 14 at 25 °C)—that’s exactly what the AP asks you to calculate for strong acid/base problems (Topic 8.2). For extra practice and reminders, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and the AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate pOH first and then convert to pH for strong base problems?
For a strong base you first get [OH−] from the formula and then use logs. Steps: 1. Write [OH−]. For group I hydroxides (NaOH, KOH) [OH−] = initial molarity. For a group II hydroxide (Ca(OH)2, Ba(OH)2) [OH−] = 2 × initial molarity (each formula unit gives 2 OH−). Example: 0.020 M NaOH → [OH−] = 0.020 M. 0.020 M Ca(OH)2 → [OH−] = 0.040 M. 2. Calculate pOH: pOH = −log[OH−]. Example: pOH = −log(0.020) = 1.70. 3. Convert to pH using Kw at 25 °C: pH = 14.00 − pOH. Example: pH = 14.00 − 1.70 = 12.30. On the AP exam you’re expected to use complete dissociation and these formulas to calculate pOH and pH (CED 8.2.A). For extra practice, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and lots of problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we say strong acids and bases are 100% ionized in solution?
We say strong acids and bases are “100% ionized” because in water their acid or base dissociation equilibrium lies so far to the right that essentially all original molecules are converted to ions. For a strong acid (HCl, HNO3, HBr, HClO4, HI) the reaction HA + H2O → H3O+ + A− has a Ka so large (≫1) that [H3O+] ≈ [initial HA]; AP CED 8.2.A.1 uses this to calculate pH directly from the starting molarity. For strong bases (group 1 hydroxides, and group 2 giving 2 OH− per formula unit like Ca(OH)2) the solid dissociates completely so [OH−] equals the starting concentration (or twice it for group-2), so you calculate pOH/pH easily (CED 8.2.A.2). Practically, the tiny fraction left undissociated is negligible compared to measurement precision and water’s autoionization. For practice applying this idea, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and more problems at https://library.fiveable.me/practice/ap-chemistry.
What's the relationship between [H+] and [OH-] in any aqueous solution at 25°C?
At 25°C the product of the hydronium and hydroxide concentrations is constant: [H+][OH−] = Kw = 1.0 × 10^−14. That means if you know one, you can find the other: [OH−] = Kw / [H+] (or [H+] = Kw / [OH−]). In log form this gives pH + pOH = 14 (since pH = −log[H+] and pOH = −log[OH−]). For strong acids/bases (Topic 8.2), complete ionization means the acid’s initial molarity equals [H+] or the base’s dissociation gives [OH−], so you can directly calculate pH or pOH and then get the other using Kw or pH+pOH=14. For more AP-aligned review and worked examples, see the Topic 8.2 study guide (https://library.fiveable.me/ap-chemistry/unit-8/ph-poh-strong-acids-bases/study-guide/AhVlrEQS1kkfZGGWdFNT) and practice problems (https://library.fiveable.me/practice/ap-chemistry).