Skills you'll gain in this topic:
- Understand how water's polarity and hydrogen bonds create unique properties.
- Discuss how cohesion, adhesion, and surface tension support biological processes.
- Illustrate examples of water's high specific heat and temperature stability effects.
- Analyze how water's polarity and hydrogen bonding lead to cohesion, adhesion, surface tension, high specific heat, and evaporative cooling that support life.
- Connect water's properties to its essential role in supporting organisms.

💧Water
Water is a polar molecule, meaning the ends have opposite partial charges or unequal distribution of charge. The chemical formula of water (H₂O) tells us that a water molecule is made up of two hydrogens and one oxygen.
Why is water polar?
- Hydrogen atoms carry partial positive charges (δ+)
- Oxygen atom has a partial negative charge (δ-)
- This unequal distribution of charge creates a molecule with distinct positive and negative regions
- Water is polar because the covalent bonds between oxygen and hydrogen are polar covalent bonds: oxygen attracts the shared electrons more strongly than hydrogen, producing a partial negative charge on oxygen and partial positive charges on the hydrogens.
The simple phrase, "like attracts like," helps us to remember that polar substances are attracted to other polar substances, just as nonpolar substances are attracted to other nonpolar substances.
Examples of polarity in everyday life:
- Ever tried mixing oil and water? They separate because:
- Oil molecules are nonpolar with equally balanced charges
- Water molecules are polar with positive and negative poles
- This makes oil and water immiscible (they do not combine)
- Polar and nonpolar substances don't attract because they are not alike
In regard to water, there is specific terminology to describe attractive interactions:
- Hydrophilic substances have an affinity for water, which means they attract water
- Hydrophobic substances avoid water or do not have an affinity for water
- Example: Lipids are hydrophobic because they have relatively nonpolar bonds, while water is polar
Water is the most common molecule in living organisms, so it is important that we understand its interactions.
Hydrogen Bonding

Hydrogen bonding is a type of chemical intermolecular bond that occurs when a hydrogen atom bonds to a highly electronegative atom, including oxygen, nitrogen, and fluorine. In water, each O–H bond is a polar covalent bond, which creates partial charges and makes the molecule polar. Because water molecules are polar, the partial positive hydrogen of one water molecule is attracted to the partial negative oxygen of a neighboring water molecule, forming a hydrogen bond between molecules. Hydrogen bonds are generally weaker than covalent bonds, but they are still relatively strong and play an important role in many chemical and biological processes.
In biology, hydrogen bonding is critical, as hydrogen bonding occurs in many different types of molecules, including water, DNA, and proteins. Hydrogen bonding is responsible for many molecular properties, such as the shape and function of proteins, the stability of many chemical compounds, and the formation of intermolecular interactions in crystalline solids. Hydrogen bonds between water molecules give water the properties of cohesion, adhesion, surface tension, specific heat, and evaporative cooling.
Properties of Water
Image courtesy of ABCteach.1. Cohesion: the attraction of water molecules. Strong cohesive forces are present because they form hydrogen bonds with each other.
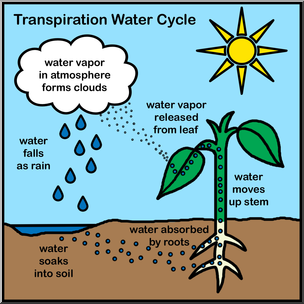
- Example: Cohesion due to hydrogen bonding contributes to the transport of water and nutrients against gravity in plants. Transpiration is the loss of water from a plant in the form of water vapor.
2. Adhesion: when one substance is attracted to another. Water adheres to other molecules and surfaces.
- Example: water on a glass surface, like a window or windshield.
3. Surface Tension: difficulty to break the surface of the water because of cohesive forces.
- Example: water strider insects rely on surface tension to stay afloat and walk on the surface of bodies of freshwater.
4. Specific Heat: the amount of heat energy required to raise 1 gram of a substance by 1°C. Water has a high specific heat capacity, meaning it can absorb or release a large amount of heat with only a small change in its own temperature. In living organisms, this helps maintain homeostatic body temperature because water resists sudden temperature changes. Large bodies of water also change temperature slowly, which helps stabilize aquatic environments.
5. Evaporative Cooling: water has a high heat of vaporization, so the water can absorb a lot of heat and leave the surface cooler. In living organisms, this property allows for body temperature to be maintained.
- Example: excess body heat is used to convert beads of sweat/perspiration into vapor, which cools down the body
Water's special properties make it essential for all living things. Its polarity and hydrogen bonding create unique abilities that support life in countless ways. Through cohesion, adhesion, surface tension, high specific heat, and evaporative cooling, water helps create stable environments inside cells and throughout ecosystems. When you see water in your everyday life - whether it's rain falling, sweat cooling your body, or helping plants grow - you're witnessing these properties in action. As you continue studying biology, you'll see how water connects to almost every topic you'll learn about. The more you understand about water's structure and behavior, the better you'll understand how life works!
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
adhesion | The attractive force between water molecules and other polar substances, allowing water to stick to different surfaces. |
cohesion | The attractive force between water molecules that causes them to stick together, resulting from hydrogen bonding. |
evaporative cooling | The process by which the evaporation of water removes heat from an organism or environment. |
heat of vaporization | The energy required to convert a liquid to a gas, enabling water to remove heat from organisms through evaporative cooling. |
homeostasis | The maintenance of stable internal environmental conditions in an organism despite external and internal changes. |
hydrogen bond | Weak attractive forces between a hydrogen atom bonded to an electronegative atom and another electronegative atom, occurring between or within biological molecules. |
polar covalent bonds | Chemical bonds between atoms where electrons are unequally shared, resulting in partial positive and negative charges. |
polarity | The unequal distribution of electrical charge in a molecule, resulting in one end being partially positive and the other partially negative. |
specific heat capacity | The amount of energy required to raise the temperature of a substance by one degree, allowing water to resist rapid temperature changes. |
surface tension | The property of water that allows its surface to resist breaking, resulting from hydrogen bonding between adjacent water molecules. |
Frequently Asked Questions
What is a polar covalent bond and how does it form in water molecules?
A polar covalent bond is a covalent bond where the electrons are shared unequally because one atom is more electronegative. In H2O, oxygen is much more electronegative than hydrogen, so the shared electrons spend more time near oxygen. That gives oxygen a partial negative charge (δ–) and each hydrogen a partial positive charge (δ+). Because the molecule is bent (∼104.5°), these partial charges don’t cancel, creating a dipole moment—the molecule has a positive end and a negative end. Those δ+ hydrogens on one water can form hydrogen bonds with the δ– oxygen on another water molecule, producing cohesion, adhesion, and high specific heat (all AP CED keywords). This idea is tested on the AP, so review the Topic 1.1 study guide on Fiveable (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and try practice questions (https://library.fiveable.me/practice/ap-biology).
Why is water considered polar and what does that actually mean?
Water is polar because the oxygen atom is much more electronegative than hydrogen, so the O–H bonds are polar covalent: electrons sit closer to O, giving O a partial negative charge (δ–) and each H a partial positive charge (δ+). Because the molecule is bent (not linear), those partial charges don’t cancel—the molecule has a net dipole moment. That polarity lets each water molecule form hydrogen bonds (weak attractions) between the δ+ H of one molecule and the δ– O of another. Biological consequences (what the AP CED expects you to know): hydrogen bonding causes cohesion, adhesion, and high surface tension; water’s polarity makes it an excellent solvent for other polar/ionic molecules; and it gives water high specific heat and high heat of vaporization, important for thermal regulation and evaporative cooling. For a concise AP-aligned review, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and more unit resources (https://library.fiveable.me/ap-biology/unit-1).
How do hydrogen bonds form between water molecules?
Hydrogen bonds form between water molecules because of water’s polar covalent bonds and bent shape. Oxygen is more electronegative than hydrogen, so each O–H bond pulls electron density toward oxygen, giving oxygen a partial negative charge (δ–) and each hydrogen a partial positive charge (δ+). Because the H–O–H molecule is bent, the dipole moment isn’t cancelled, so one water molecule’s δ+ hydrogen is electrostatically attracted to a neighboring molecule’s δ– oxygen. That weak, noncovalent attraction is a hydrogen bond. These H-bonds are individually weak but numerous, producing cohesion, adhesion, high surface tension, high specific heat, and high heat of vaporization—all listed in the CED (LO 1.1.A.1–1.1.A.2). For AP prep, be ready to explain how polarity (δ+/δ–), molecular geometry (bent), and hydrogen bonding link to those properties. Want a concise topic review and practice Qs? Check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and find 1,000+ practice problems at (https://library.fiveable.me/practice/ap-biology).
What's the difference between hydrogen bonds and regular covalent bonds?
Covalent bonds are strong bonds where atoms share electrons to form molecules—in water, the O–H bonds are polar covalent because oxygen is more electronegative than hydrogen, so electrons spend more time near O, creating partial charges (δ– on O, δ+ on H). Hydrogen bonds are weak, noncovalent attractions between a hydrogen that’s already covalently bonded to an electronegative atom (like O) and another electronegative atom (often O or N) on a nearby molecule. Key differences: strength (covalent >> hydrogen), location (within a molecule vs. between molecules or different parts of a large molecule), and function (covalent defines molecular structure; hydrogen bonds give water properties like cohesion, adhesion, surface tension, high specific heat and evaporative cooling). On the AP exam expect you to use terms like polar covalent, electronegativity, partial charges, dipole moment, and relate hydrogen bonding to biological functions (CED 1.1.A.1–1.1.A.2). For a quick review, see the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and practice questions (https://library.fiveable.me/practice/ap-biology).
I'm confused about cohesion vs adhesion - can someone explain the difference?
Cohesion and adhesion both come from water’s polarity and hydrogen bonds, but they’re different: cohesion is water sticking to water (H bonds between H2O molecules)—that’s what creates surface tension and helps a water column stay unbroken in xylem (cohesion-tension theory). Adhesion is water sticking to other polar surfaces (like cellulose in plant cell walls or glass)—that helps capillary action, where water climbs narrow tubes because it’s attracted to the tube walls. Think: cohesion = water-to-water; adhesion = water-to-something-else. Both are listed in the CED under 1.1.A.2 and are common AP exam topics (MCQs or FRQs about surface tension, capillary action, or plant water transport). For a quick review, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj). Want more practice? Fiveable has tons of AP Bio practice questions (https://library.fiveable.me/practice/ap-biology).
Why does water have such a high specific heat capacity compared to other liquids?
Water’s high specific heat comes from its polarity and the hydrogen bonds between molecules. Each water molecule can form up to four H-bonds with neighbors (because O is highly electronegative and the H’s are δ+), so when you heat water much of the added energy goes into breaking or loosening those H-bonds rather than raising kinetic energy (temperature). That means you need more energy per gram to change water’s temperature compared with liquids that lack extensive H-bonding. Biologically, this stabilizes internal and environmental temperatures (helps homeostasis and evaporative cooling)—exactly the CED points about polarity, hydrogen bonding, and thermal regulation (1.1.A.1–ii, iii). For quick review, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and practice problems at (https://library.fiveable.me/practice/ap-biology) to see AP-style questions on these concepts.
How does water's high heat of vaporization help with temperature regulation in living things?
Because hydrogen bonds hold water molecules together, water has a high heat of vaporization—you must input a lot of energy to turn liquid water into gas. In living things that becomes evaporative cooling: when surface water (like sweat or water on leaves) evaporates, it takes that large amount of heat energy with it, lowering the temperature of the remaining tissues and helping maintain homeostasis. That’s why sweating cools you efficiently and why transpiration helps plants avoid overheating. On the AP Bio exam, connect this property back to polarity and hydrogen bonding (CED 1.1.A.1 and 1.1.A.1.iii) when explaining thermal regulation. For a quick review of these links between water’s bonding and biological function, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj). For extra practice on exam-style questions, try the AP Bio practice set (https://library.fiveable.me/practice/ap-biology).
What causes surface tension in water and why can some insects walk on water?
Surface tension in water comes from cohesion between water molecules: oxygen is highly electronegative, so each H–O bond is polar (partial charges δ+ on H, δ– on O). Those partial charges let molecules form hydrogen bonds with neighbors, creating a “skin” at the air–water interface that resists stretching or breaking (this is exactly the cohesion/surface tension idea in CED LO 1.1.A and 1.1.A.2). Some insects (like water striders) can walk on water because their weight is small and their long, hydrophobic legs distribute force so they don’t break that surface “skin.” The surface tension supports the insect by producing an upward force where the surface is slightly depressed; hydrophobic legs also reduce wetting, helping keep the surface intact. For more AP-aligned review, see the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj), the Unit 1 overview (https://library.fiveable.me/ap-biology/unit-1), and extra practice (https://library.fiveable.me/practice/ap-biology).
Why is water's polarity so important for biological processes?
Polarity gives each water molecule partial charges (δ– on O, δ+ on H) because O is more electronegative; those polar covalent bonds let water form hydrogen bonds with other polar molecules. That’s huge for biology: hydrogen bonding causes cohesion, adhesion, and high surface tension (helps xylem transport and insect walkers), and makes water an excellent solvent for ions and polar molecules (so nutrients, salts, and proteins dissolve and interact). Water’s high specific heat and high heat of vaporization (both products of hydrogen bonds) buffer temperature changes and enable evaporative cooling—key for homeostasis. Polarity also drives hydrophobic vs. hydrophilic interactions that fold proteins and assemble membranes. These are exactly the CED ideas in LO 1.1.A (structure → properties → biological function). For a focused review and practice problems tied to these concepts, see the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj), the Unit 1 overview (https://library.fiveable.me/ap-biology/unit-1), and AP practice questions (https://library.fiveable.me/practice/ap-biology).
How do hydrogen bonds between water molecules affect the properties of water?
Hydrogen bonds between water molecules—formed because O is more electronegative than H, giving partial charges (δ– on O, δ+ on H)—create most of water’s key properties that biology depends on (LO 1.1.A). Cohesion: H-bonds hold molecules together, enabling water transport in plants (cohesion-tension, high surface tension). Adhesion: water sticks to other polar surfaces, helping capillary action. Thermal properties: lots of H-bonds mean high specific heat and high heat of vaporization, so water buffers temperature changes and enables evaporative cooling (thermal regulation/homeostasis). Solvent behavior: the polar nature and H-bonding make water the “solvent of life,” dissolving hydrophilic molecules while excluding hydrophobic ones, which drives macromolecule folding and membrane formation. These are exactly the CED keywords you should know for exam questions about water’s biological functions. For a quick review, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj); for unit review or practice Qs, see the Unit 1 page (https://library.fiveable.me/ap-biology/unit-1) and practice problems (https://library.fiveable.me/practice/ap-biology).
I don't understand how water helps maintain homeostasis in organisms - can someone explain?
Think of water as a built-in temperature buffer and transport medium in your body. Because H2O is polar (O is δ–, H is δ+), molecules form hydrogen bonds with each other and with other polar molecules. Those H-bonds give water a high specific heat—it takes a lot of energy to change temperature—so bodies (and environments) stay near constant temps (helps maintain homeostasis). Water’s high heat of vaporization enables evaporative cooling (sweating/ panting) to remove heat efficiently. Hydrogen bonds also cause cohesion and adhesion, producing surface tension and capillary action that move water through plants and blood vessels. On the AP exam you’ll be asked to EXPLAIN these links (LO 1.1.A)—connect polarity → hydrogen bonding → the property → the biological function. For a quick review, see the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and more unit review/practice problems at (https://library.fiveable.me/ap-biology/unit-1) and (https://library.fiveable.me/practice/ap-biology).
What happens when water evaporates and how does this cool down the environment?
When water evaporates, hydrogen bonds between water molecules must be broken so individual molecules can escape as gas. Breaking those bonds requires a lot of energy (water’s high heat of vaporization), so the molecules that actually evaporate are the ones with the highest kinetic energy. When those high-energy molecules leave, the average kinetic energy of the remaining liquid drops—that means the liquid (and nearby surfaces/air) cools. That’s evaporative cooling in biology: sweating or panting removes high-energy water, helping maintain body temperature (homeostasis). This is exactly tied to CED 1.1.A.1 (water’s high specific heat and high heat of vaporization). You might see this concept on multiple-choice or free-response questions asking how water’s polarity and hydrogen bonding affect thermal regulation. For a focused review, see the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj), the unit overview (https://library.fiveable.me/ap-biology/unit-1), and more practice problems (https://library.fiveable.me/practice/ap-biology).
Why do we need to know about water's molecular structure for AP Bio?
You need water’s molecular structure because a lot of the AP Bio ideas and questions flow directly from it. The bent shape and polar covalent O–H bonds give water partial charges (δ+ on H, δ– on O), so water forms hydrogen bonds. Those H-bonds explain cohesion, adhesion, surface tension, high specific heat, and high heat of vaporization—all listed in LO 1.1.A of the CED—and those properties underlie thermoregulation, capillary action, evaporative cooling, and why water is the “solvent of life.” On the exam you’ll be asked to explain how these properties affect biological function (multiple-choice and FRQ Concept Explanation/Visual Representation tasks); Unit 1 is 8–11% of the exam. If you want a concise review tied to the CED keywords (dipole, hydrophilic vs hydrophobic, cohesion-tension theory), check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) and practice lots of problems (https://library.fiveable.me/practice/ap-biology).
How are hydrogen bonds different from the bonds that hold H and O together in a water molecule?
Inside a water molecule, the H–O connections are polar covalent bonds: oxygen is more electronegative than hydrogen, so the shared electrons sit closer to O, giving O a partial negative (δ–) and each H a partial positive (δ+). Those bonds hold the atoms together (intramolecular) and are relatively strong. Hydrogen bonds are intermolecular attractions between the δ+ hydrogen on one water molecule and the δ– oxygen on a neighboring water molecule. They’re much weaker than covalent bonds (they form and break easily), but because each water molecule can form up to four H-bonds, these interactions create cohesion, high specific heat, high heat of vaporization, surface tension, and capillary action—all key to biological function (CED LO 1.1.A; keywords: polar covalent bond, electronegativity, δ+ / δ–, hydrogen bond). Want practice or a quick refresher? Check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj) or the Unit 1 overview (https://library.fiveable.me/ap-biology/unit-1). For extra practice problems, go here: (https://library.fiveable.me/practice/ap-biology).
What are some real examples of how water's properties help living organisms survive?
Water’s polarity and hydrogen bonding let it do a lot that keeps organisms alive. Examples you should remember for the AP: - Thermal stability: High specific heat means water buffers temperature changes, helping organisms maintain homeostasis (e.g., body temperature regulation in humans). - Evaporative cooling: High heat of vaporization lets sweating or panting remove lots of heat, keeping body temp steady. - Cohesion and adhesion: Hydrogen bonds cause cohesion (water molecules stick together) and adhesion (stick to cell walls), enabling capillary action—how water moves up xylem in plants (cohesion-tension theory). - Surface tension: Strong surface tension lets insects like water striders walk on water and supports thin water films in lungs. - Universal solvent: Polarity dissolves many solutes so nutrients, gases, and ions move and react in cells; hydrophilic vs. hydrophobic interactions also drive protein folding and membrane formation. These points map to LO 1.1.A in the CED. For a quick review, check the Topic 1.1 study guide (https://library.fiveable.me/ap-biology/unit-1/structure-water-hydrogen-bonding/study-guide/bMEKm5Pi5y3Y3XRln0Bj). For broader Unit 1 review or practice problems, see (https://library.fiveable.me/ap-biology/unit-1) and (https://library.fiveable.me/practice/ap-biology).