Principles of potentiometry
Fundamentals of potentiometric measurements
Potentiometry is an electroanalytical method that measures the potential difference between two electrodes immersed in a sample solution: a reference electrode (which provides a stable, known potential) and an indicator electrode (which responds to the analyte). The measured potential is directly related to the activity of a specific analyte in solution.
The relationship between measured potential and analyte activity is described by the Nernst equation:
- = measured cell potential
- = standard electrode potential
- = gas constant (8.314 J/mol·K)
- = temperature in Kelvin
- = number of electrons transferred (or charge of the ion)
- = Faraday's constant (96,485 C/mol)
- = activity of the analyte
At 25°C, this simplifies to the more commonly used form:
One practical detail worth remembering: potentiometric measurements require a high-impedance voltmeter (or pH meter). This keeps current flow essentially at zero, which prevents the measurement itself from shifting the equilibrium and changing the potential you're trying to read.
Applications of potentiometry
- pH determination using a glass electrode, the most common potentiometric measurement
- Ion analysis using ion-selective electrodes (ISEs) tailored to specific targets (e.g., sodium ISE, calcium ISE, fluoride ISE)
- Redox measurements using inert metal electrodes (like platinum) to monitor oxidation-reduction potential (ORP)
- Potentiometric titrations, where you track the potential as a function of titrant volume added. Instead of relying on a color indicator, you monitor the potential curve and identify the endpoint from the inflection point (the steepest part of the curve). These are commonly used in acid-base titrations (with a pH electrode) and precipitation titrations (with a silver ISE for halide determination).
Ion-selective electrode construction
Components of an ion-selective electrode
An ISE has four main components that work together:
- Ion-selective membrane: The critical component that gives the electrode its selectivity. The membrane material depends on the target ion:
- Glass membranes for pH electrodes (-responsive glass)
- Crystalline membranes like for fluoride ISEs
- Polymer (liquid) membranes containing ionophores (selective binding molecules) for ions like or
- Internal reference solution: A solution with a fixed, known concentration of the target ion. This keeps the potential at the inner membrane surface constant, so any change in the measured potential comes only from the sample side.
- Internal reference electrode: Typically immersed in the internal reference solution. It provides a stable electrical contact to the measuring circuit.
- External reference electrode: A separate electrode (often a double-junction design) that provides a stable potential independent of sample composition. The double junction helps prevent contamination of the reference electrolyte by the sample.
Functioning of an ion-selective electrode
When the ISE contacts a sample containing the target ion, a potential difference develops across the membrane. This happens because the activity of the target ion differs between the sample side and the internal reference solution side. The membrane selectively allows interaction with the target ion, generating a measurable voltage.
That voltage is then related back to concentration using the Nernst equation. The selectivity of the measurement depends entirely on how well the membrane discriminates between the target ion and other ions present in the sample. A perfectly selective membrane would respond only to the target ion, but in practice, some degree of interference from other ions always exists.
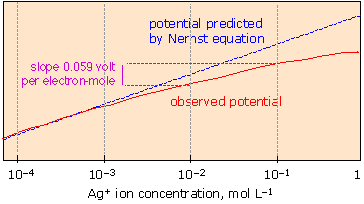
Analysis of potentiometric data
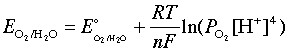
Calibration curves and concentration determination
To determine unknown concentrations, you first build a calibration curve:
- Prepare a series of standard solutions with known concentrations of the target ion (typically spanning several orders of magnitude).
- Measure the potential for each standard using the ISE and reference electrode.
- Plot the measured potential (y-axis) against (x-axis). The Nernst equation predicts this should be linear.
- Check the slope. At 25°C, the theoretical (Nernstian) slope is mV per decade of concentration, where is the charge on the ion. For a monovalent ion like , that's 59.16 mV/decade; for a divalent ion like , it's 29.58 mV/decade.
- Measure the potential of your unknown sample and use the calibration curve to find its concentration, either by interpolation from the graph or by plugging values into the Nernst equation.
If your experimental slope deviates significantly from the theoretical value, something is off. Common culprits include an aging or damaged membrane, poor temperature control, or contaminated standards.
Quantifying selectivity and detection limits
Selectivity coefficients () quantify how much an interfering ion affects the ISE's response to the target ion . A smaller means better selectivity. For example, if , the interfering ion has only 1/1000th the effect of the target ion at equal concentrations. These coefficients are determined experimentally, typically using the separate solution method or the fixed interference method.
The limit of detection (LOD) is the lowest target ion concentration the ISE can reliably distinguish from background. On a calibration curve, it corresponds to the point where the linear response region begins to flatten out. Formally, it's often defined as the concentration at which the measured potential deviates from the extrapolated linear portion by a statistically meaningful amount (commonly using a 3σ criterion relative to background noise). Lower LODs are important for trace analysis, such as detecting heavy metal ions in environmental water samples.
Performance of ion-selective electrodes
Factors influencing selectivity and sensitivity
Selectivity determines whether you can trust your measurement in a real sample matrix where multiple ions are present.
- It depends on the membrane composition, the charge and ionic radius of the target vs. interfering ions, and the overall sample matrix.
- Strategies for improving selectivity: optimize membrane composition, use highly selective ionophores (molecules designed to bind preferentially to the target ion), or apply mathematical corrections using known selectivity coefficients (the Nikolsky-Eisenman equation extends the Nernst equation to account for interfering ions).
Sensitivity is the change in measured potential per unit change in analyte concentration. In Nernstian terms, you want the slope as close to the theoretical value as possible.
- Higher sensitivity means you can detect smaller concentration differences.
- Sensitivity degrades with membrane aging, temperature instability, and fouling of the membrane surface by sample components.
Strategies for improving detection limits
Reaching lower LODs matters most when analyzing trace-level analytes. Several practical strategies help:
- Optimize measurement conditions: Control temperature carefully (the Nernst slope is temperature-dependent), shield the system from electromagnetic interference, and allow adequate equilibration time.
- Sample pretreatment: Remove interfering ions before measurement using techniques like ion exchange chromatography or solvent extraction. This reduces the background signal that competes with the target ion response.
- Standard addition method: Instead of using an external calibration curve, spike known amounts of the target ion directly into the sample. This accounts for matrix effects because the standards experience the same matrix as the unknown. Multiple standard additions further improve accuracy.
- Membrane conditioning: Proper soaking and conditioning of the ISE membrane before use ensures a stable, reproducible response at low concentrations.